
Chromates are generally bright yellow, while dichromates are generally orange or red. Chromate and dichromate are strongly oxidizing in acidic conditions, but weakly oxidizing in basic conditions. This is to show that the subscript applies to the entire polyatomic ion. Dichromate is similar, but it contains two chromium atoms and seven oxide atoms (therefore, Cr 2 O 7 ). If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. The pK value for this reaction shows that it are often ignored at pH > 4.\): Some Polyatomic Ions Name The dichromate ion may be a somewhat weaker base than the chromate ion. Reported values vary between about −0.8 and 1.6. Let x be the chromium’s oxidation number. So, potassium ions have a total charge of +1(2) 2, whereas oxygen ions have a charge of 2(7) 14. The oxidation state of potassium is +1, while the oxidation state of oxygen is -2. The hydrogen chromate ion could also be protonated, with the formation of molecular acid, H2CrO4, but the pKa for the equilibrium Tetrahedral coordination geometries surround the chromium atoms. It is also in equilibrium with the dichromate ion:Īs a result, this equilibrium doesn’t involve a change in proton concentration, which might predict that the equilibrium is independent of pH. The hydrogen chromate ion, HCrO 4−, maybe a weak acid: Solution All polyoxyanions of chromium(VI) have structures made from tetrahedral CrO4 units sharing corners. Q 1] Explain how condensation reactions occur for chromate and dichromate ions? Oxidation of the chromium to the hexavalent form occurs, while the iron forms iron(III) oxide, Fe2O3.ĤFeCr 2O 4 + 8Na 2CO 3 + 7O 2 → 8Na 2CrO 4 + 2Fe 2O 3 + 8CO 2 Solved Examples for Chromate Formula The intermediates cover the chromium surface completely with an. In addition, chromite ore is heated with a mix of carbonate and washing soda within the presence of air. oxidation of chromium to chromate proceeds via six consecutive charge-transfer reactions. The primary chromium ore is that the mixed metal oxide chromite, FeCr2O4, found as brittle metallic black crystals or granules. 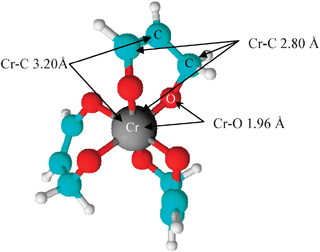
The redox potential shows that chromates are the weaker oxidant in alkaline solution than in acid solution.ĬrO 2−4 + 4 H 2O + 3 e− → Cr(OH)3 + 5 OH− 
In alkaline solution chromium(III) hydroxide is produced. In acid solution, there is the production of aquated Cr3+ ion.Ĭr 2O 2−7 + 14 H+ + 6 e− → 2Cr3+ + 7H 2O ε0 = 1.33 V Commonly three electrons are added to a chromium atom, reducing it to oxidation number +3. The chromate and dichromate ions are good strong oxidizing agents. The +2 oxidation state is very strongly reducing and that can only exist for a short time, it is very quickly oxidized to the +3 oxidation state. In the solid state, chromium also can exist in the +4 oxidation state. In aqueous solutions, chromium can exist in the +2, +3, +5, and +6 oxidation states. In solution, chromate and dichromate anions exist in equilibrium. Aqueous solutions and precipitates of chromium. But the ion of dichromate can become predominant in acidic solutions. The chromate ion is that the predominant species in alkaline solutions. The position of the equilibrium depends on both pH and therefore the analytical concentration of chromium. In a solution, chromate ions are often interconvertible. So, both chromium and oxygen atoms have six electrons in their valence. Chromium and oxygen are located at 6 and 16 groups respectively in the periodic table. Also, there is a -2 overall charge on the CrO 4 2-ion which is also should be considered to draw the lewisstructure. Chromate salt of heavy metals, lanthanides, and alkaline-earth metal metals are only very slightly soluble in water and are used as pigments. There are one chromium atom and four oxygen atoms in the chromate ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
